|
bensulfuron-methyl
Herbicide
HRAC B WSSA 2; sulfonylurea
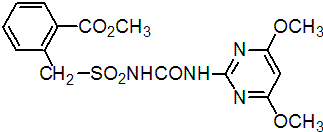
NOMENCLATURE
bensulfuron-methyl
Common name bensulfuron-methyl
IUPAC name methyl a-(4,6-dimethoxypyrimidin-2-ylcarbamoylsulfamoyl)-o-toluate
Chemical Abstracts name methyl 2-[[[[[(4,6-dimethoxypyrimidin-2-yl)-amino]carbonyl]amino]sulfonyl]methyl]benzoate
CAS RN [83055-99-6] EEC no. 401-340-6 (ELINCS) Development codes DPX-F5384 (DuPont)
bensulfuron
Common name bensulfuron (BSI, ANSI, WSSA, draft E-ISO, (m) draft F-ISO)
IUPAC name a-(4,6-dimethoxypyrimidin-2-ylcarbamoylsulfamoyl)-o-toluic acid
Chemical Abstracts name 2-[[[[[(4,6-dimethoxy-2-pyrimidinyl)amino]carbonyl]amino]sulfonyl]methyl]benzoic acid
CAS RN [99283-01-9]
PHYSICAL CHEMISTRY
bensulfuron-methyl
Composition Tech. is 97.5%. Mol. wt. 410.4 M.f. C16H18N4O7S Form White odourless solid. M.p. 185-188 ºC; tech. 179.4 °C V.p. 2.8 ´ 10-9 mPa (25 ºC) KOW logP = -0.99 (pH 5), 0.79 (pH 7), 2.18 (pH 9) (all 25 ºC) Henry 2 ´ 10-11 Pa m3 mol-1 (calc.) S.g./density 1.49 (20 °C) Solubility In water 2.1 (pH 5), 67 (pH 7), 3100 (pH 9) (all in mg/l, 25 ºC). In acetone 5.10, acetonitrile 3.75, dichloromethane 18.4, ethyl acetate 1.75, n-heptane 3.62 ´ 10-4, xylene: 0.229 (all in g/l, 20 ºC). Stability Aqueous solutions are most stable under slightly alkaline conditions (pH 8), and slowly degrade under acidic conditions; DT50 6 d (pH 4), stable (pH 7), 141 d (pH 9) (all 25 ºC). pKa 5.2
bensulfuron
Mol. wt. 396.4 M.f. C15H16N4O7S
COMMERCIALISATION
History Herbicidal activity of bensulfuron-methyl reported by T. Yayama et al. (Proc. 9th Asian Pacific Weed Sci. Soc. Congr., 1983, p. 554). Introduced by E. I. du Pont de Nemours and Co.; first approval in 1985. Patents US 4420325 Manufacturers DuPont; Sannong; Sharda; Shenyang; Sundat
APPLICATIONS
bensulfuron-methyl
Biochemistry Branched chain amino acid synthesis (ALS or AHAS) inhibitor. Acts by inhibiting biosynthesis of the essential amino acids valine and isoleucine, hence stopping cell division and plant growth. Selectivity is due to rapid metabolism in the crop. Metabolic basis of selectivity reviewed (M. K. Koeppe & H. M. Brown, Agro-Food-Industry, 6, 9-14 (1995)). Mode of action Selective systemic herbicide, absorbed by the foliage and roots, with rapid translocation to the meristematic tissues. Uses Selective pre- and post-emergence control of annual and perennial weeds and sedges (e.g. Butomus umbellatus, Scirpus maritimus, Scirpus mucronatus, Alisma plantago-aquatica, Sparganium erectum, Cyperus spp., Typha spp., etc.) in flooded or wetland rice, at 46-60 g/ha. Formulation types WG; WP. Selected products: 'Londax' (DuPont); mixtures: 'Fujigrass' (+ esprocarb) (DuPont, Syngenta); 'Zark' (+ mefenacet) (DuPont, Kumiai, Nihon Bayer, Sankyo Agro); 'Innova GR 75' (+ fentrazamide) (Nihon Bayer); 'Kusamets' (+ thenylchlor) (Tokuyama, Hokko, Nihon Nohyaku); 'Kusastop' (+ indanofan) (Nihon Nohyaku); 'Patful' (+ oxaziclomefone+ pyriminobac-methyl) (Kumiai); 'Prosper' (+ mefenacet+ pyriminobac-methyl) (Kumiai); 'Topgun' (+ bromobutide+ pentoxazone+ pyriminobac-methyl) (Kumiai); 'Wolf-Ace' (+ mefenacet+ thiobencarb) (Kumiai)
OTHER PRODUCTS
bensulfuron-methyl
'Rozal' (DuPont); 'Escuri' (Crystal) mixtures: 'Kingdom' (+ pyributicarb+ thenylchlor) (DuPont); 'Kusanain' (+ dimethametryn+ pretilachlor+ pyributicarb) (DuPont); 'Sindax' (+ metsulfuron-methyl) (DuPont); 'Bazooka 36' (+ cyhalofop-butyl+ thenylchlor+ azimsulfuron) (Nihon Nohyaku); 'Bazooka A' (+ cyhalofop-butyl+ azimsulfuron) (Nihon Nohyaku, Hokko, Tokuyama, DuPont); 'Bazooka' (+ cyhalofop-butyl+ thenylchlor) (Nihon Nohyaku, Hokko, Tokuyama, DuPont); 'Cao Neng' (+ quinclorac) (Anhui); 'Dancing A' (+ indanofan+ azimsulfuron) (Nihon Nohyaku); 'Dancing L' (+ indanofan) (Nihon Nohyaku); 'Dancing-Power A' (+ clomeprop+ indanofan+ azimsulfuron) (Nihon Nohyaku); 'Dancing-Power L' (+ clomeprop+ indanofan) (Nihon Nohyaku); 'Dinaman' (+ clomeprop+ indanofan) (Nihon Nohyaku); 'Duet EDF' (+ propanil) (Riceco); 'Homerun' (+ oxaziclomefone) (Hokko); 'Inebrite' (+ fentrazamide+ azimsulfuron) (Bayer CropScience); 'Inegreen D' (+ cyhalofop-butyl+ daimuron+ mefenacet) (Nihon Bayer, Hokko); 'Inegreen' (+ cyhalofop-butyl+ mefenacet) (Nihon Bayer); 'Innova GR 51' (+ daimuron+ fentrazamide) (Nihon Bayer); 'Innova L SC' (+ daimuron+ fentrazamide) (Nihon Bayer); 'Innova SC' (+ fentrazamide) (Nihon Bayer); 'Joystar' (+ cafenstrole+ cyhalofop-butyl+ daimuron) (Kumiai); 'Joyster' (+ cafenstrole+ cyhalofop-butyl+ daimuron) (Kumiai); 'Karshot' (+ pyributicarb) (Sankyo Agro); 'Kusamets FL' (+ thenylchlor) (Nihon Nohyaku, Tokuyama, Hokko); 'Kusastop A' (+ indanofan+ azimsulfuron) (Nihon Nohyaku); 'Kusatory Ace Jumbo' (+ cafenstrole+ daimuron) (Sankyo Agro); 'Kusatory E Jumbo' (+ pyributicarb) (Sankyo Agro); 'Londanil' (+ propanil) (Crystal); 'Masakari A Jumbo' (+ clomeprop+ indanofan+ azimsulfuron) (Nihon Nohyaku); 'Masakari L Jumbo' (+ clomeprop+ indanofan) (Nihon Nohyaku); 'Non-Poong' (+ benfuresate) (Bayer CropScience); 'Papika A I Kilo' (+ cyhalofop-butyl+ thenylchlor+ azimsulfuron) (Nihon Nohyaku, Hokko, Tokuyama, Dow AgroSciences, DuPont); 'Papika A' (+ cyhalofop-butyl+ thenylchlor+ azimsulfuron) (Tokuyama, Nihon Nohyaku, Hokko, DuPont, Dow AgroSciences); 'Papika' (+ cyhalofop-butyl+ thenylchlor) (Tokuyama, Nihon Nohyaku, Hokko, DuPont, Dow AgroSciences); 'Pro-Pack 80EDF' (+ propanil) (Agriliance); 'Push' (+ dimepiperate) (Hokko, Bayer CropScience, DuPont); 'Rakudar' (+ cafenstrole+ daimuron) (Sankyo Agro); 'Ranger' (+ dimepiperate+ benfuresate) (Bayer CropScience, Hokko, DuPont); 'Step' (+ benzofenap+ thenylchlor) (Nihon Nohyaku); 'Weedless A36' (+ cafenstrole+ daimuron+ azimsulfuron) (Sankyo Agro, DuPont, Eiko Kasei, SDS Biotech KK); 'Weedless' (+ cafenstrole+ daimuron) (Sankyo Agro); 'Wydori A' (+ dimethametryn+ pretilachlor+ azimsulfuron) (Nihon Nohyaku); 'Zark D' (+ daimuron+ mefenacet) (Sankyo Agro) Discontinued products: 'Mariner' * (DuPont) mixtures: 'Kusatory Jumbo' * (+ daimuron+ pyributicarb) (Sankyo); 'Tabijin A' * (+ cyhalofop-butyl+ pretilachlor+ pyriminobac-methyl+ azimsulfuron) (Kumiai)
ANALYSIS
Product and residue analysis by hplc/ms/ms (Handbook of Residue Analytical Methods, pp 400-410).
MAMMALIAN TOXICOLOGY
bensulfuron-methyl
Oral Acute oral LD50 for rats >5000 mg/kg. Skin and eye Acute percutaneous LD50 for rabbits >2000 mg/kg. Non-irritant to skin and eyes. Not a skin sensitiser (Buehler method). Inhalation LC50 (4 h) for rats 5 mg/l air. NOEL (1 y) for male dogs 21.4 mg/kg b.w. daily; reproduction (2 generation) NOEL for male rats 20 mg/kg b.w. daily; teratogenicity NOEL for rabbits 300 mg/kg b.w. daily. No developmental toxicity or teratogenicity. ADI 0.20 mg/kg. Toxicity class WHO (a.i.) U; EPA (formulation) IV EC classification R43| N; R51, R53
ECOTOXICOLOGY
bensulfuron-methyl
Birds Acute oral LD50 for mallard ducks >2510 mg/kg. Dietary LC50 (8 d) for bobwhite quail, mallard ducks >5620 mg/l. Fish LC50 (96 h) for rainbow trout >66, bluegill sunfish >120 mg/l. Daphnia LC50 (48 h) >130 mg/l. Algae EC50 (72 h) for Selenastrum capricornutum 0.020 mg/l. Other aquatic spp. EC50 (14 d) for Lemna gibba 0.0008 mg/l. Bees LD50 (oral) >25.7 mg/bee; (contact) >100 mg/bee. Worms LC50 >1000 mg/kg soil.
ENVIRONMENTAL FATE
Animals Almost completely biotransformed and rapidly excreted in urine and faeces of rats and goats. Plants After uptake by rice, converted to a non-herbicidal metabolite. Soil/Environment DT50 (ave.) 88.5 d on Flanagan and Keyport silt loam soils. DT50 in water 16-21 d for Italian soils.
资料收集库(DocBook) 小笨象工作室 /www.9ele.com/
|